White Paper: Protocol Design in Real-World Evidence: The Indispensable Link Between Strategic Need and Study Execution - Evidera

STaRT-RWE: structured template for planning and reporting on the implementation of real world evidence studies | The BMJ

SOPs for GCP-Compliant Clinical Trials: A Customizable Manual for Sponsors of Medical Device Trials : MS Word Template | CenterWatch
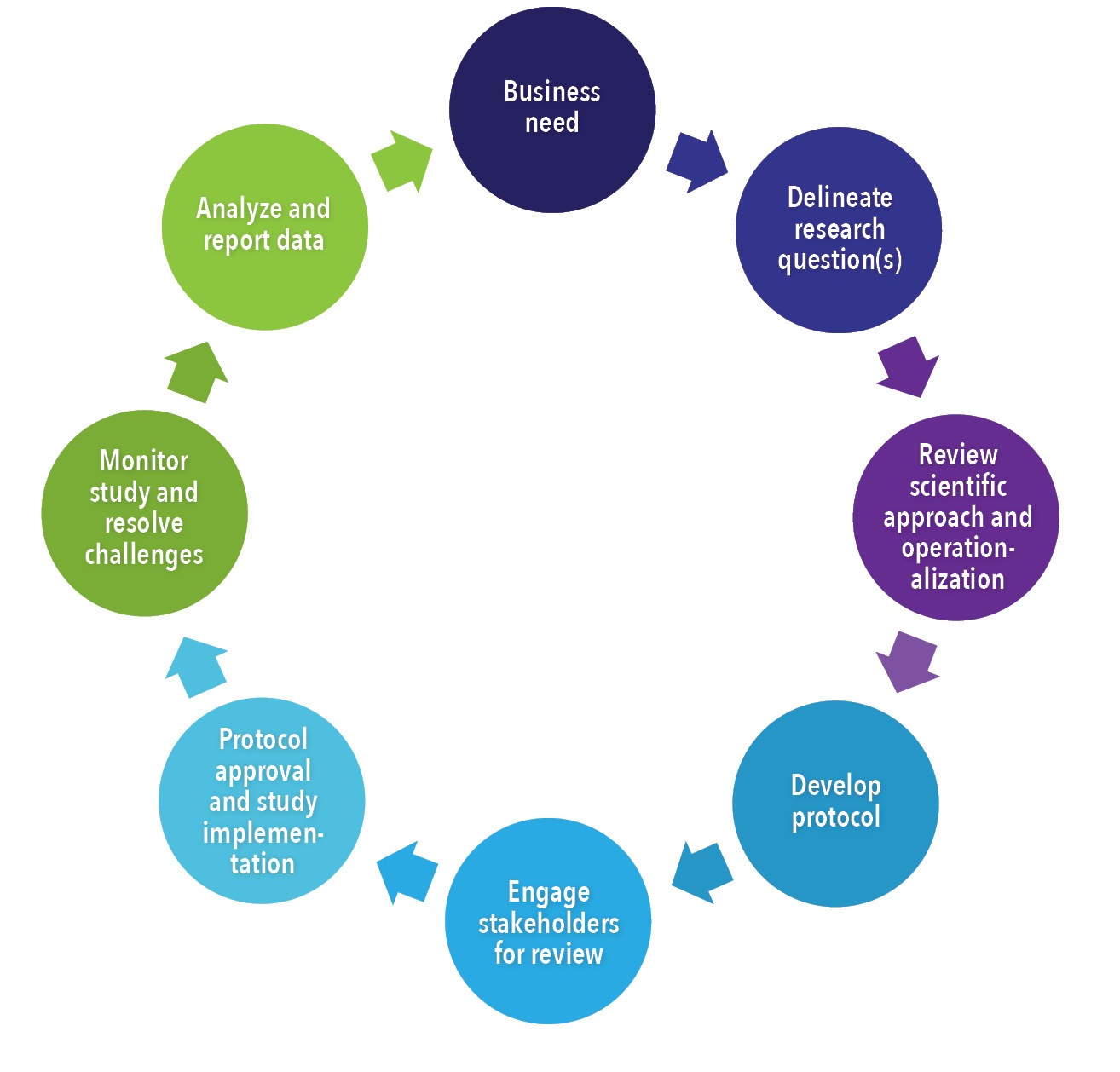
White Paper: Protocol Design in Real-World Evidence: The Indispensable Link Between Strategic Need and Study Execution - Evidera

New Template Guides Protocol Development for Medical Device Trials Using RWE | 2020-03-06 | CenterWatch