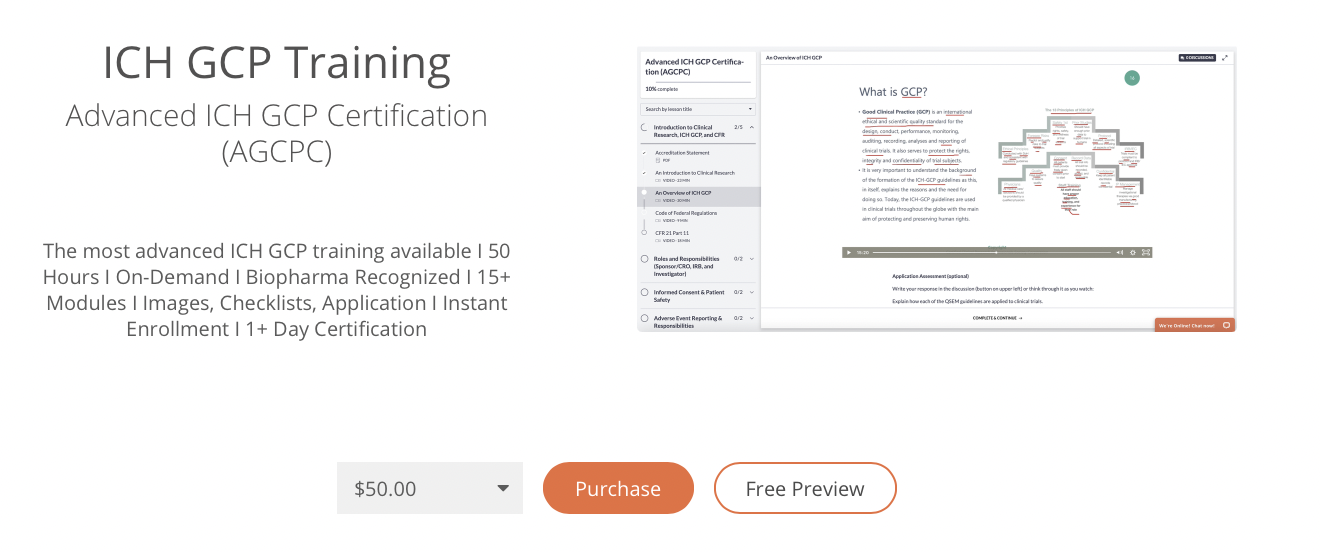
REFRESHER: ICH Good Clinical Practice (GCP) E6 (R2) and regulatory requirements for Clinical Trials (Fiona Stanley Hospital) - RETProgram

Introduction to investigators responsibilities with good clinical practice - ICH Good Clinical - StuDocu

More than a box to check: Research sponsor and clinical investigator perspectives on making GCP training relevant - ScienceDirect

PDF) Good Clinical Practice Training: Identifying Key Elements and Strategies for Increasing Training Efficiency

Good Clinical Practice: A Question & Answer Reference Guide, May 2018: Earl W. Hulihan, Earl W. Hulihan: 9780996346252: Amazon.com: Books
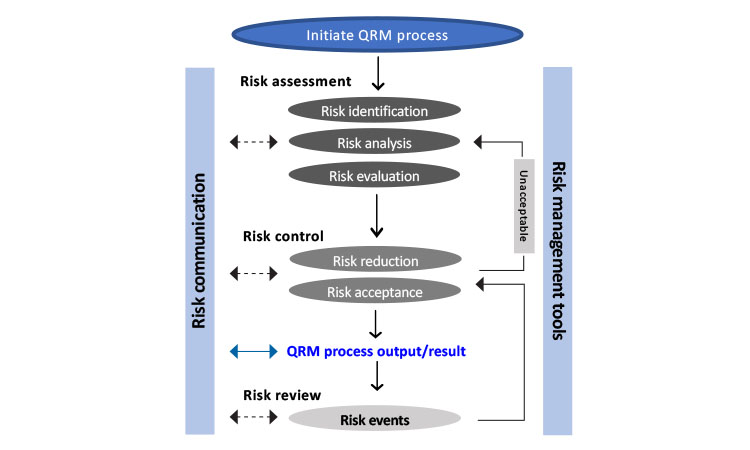
Clinical Trial Management Adaptation to ICH E6 (R2): Good Clinical Practice | Pharmaceutical Engineering
1.) Go to Website: GLOBAL HEALTH TRAINING CENTRE a. https://globalhealthtrainingcentre.tghn.org/ b. REGISTER ( TOP RIGHT CORNER)
1.) Go to Website: GLOBAL HEALTH TRAINING CENTRE a. https://globalhealthtrainingcentre.tghn.org/ b. REGISTER ( TOP RIGHT CORNER)