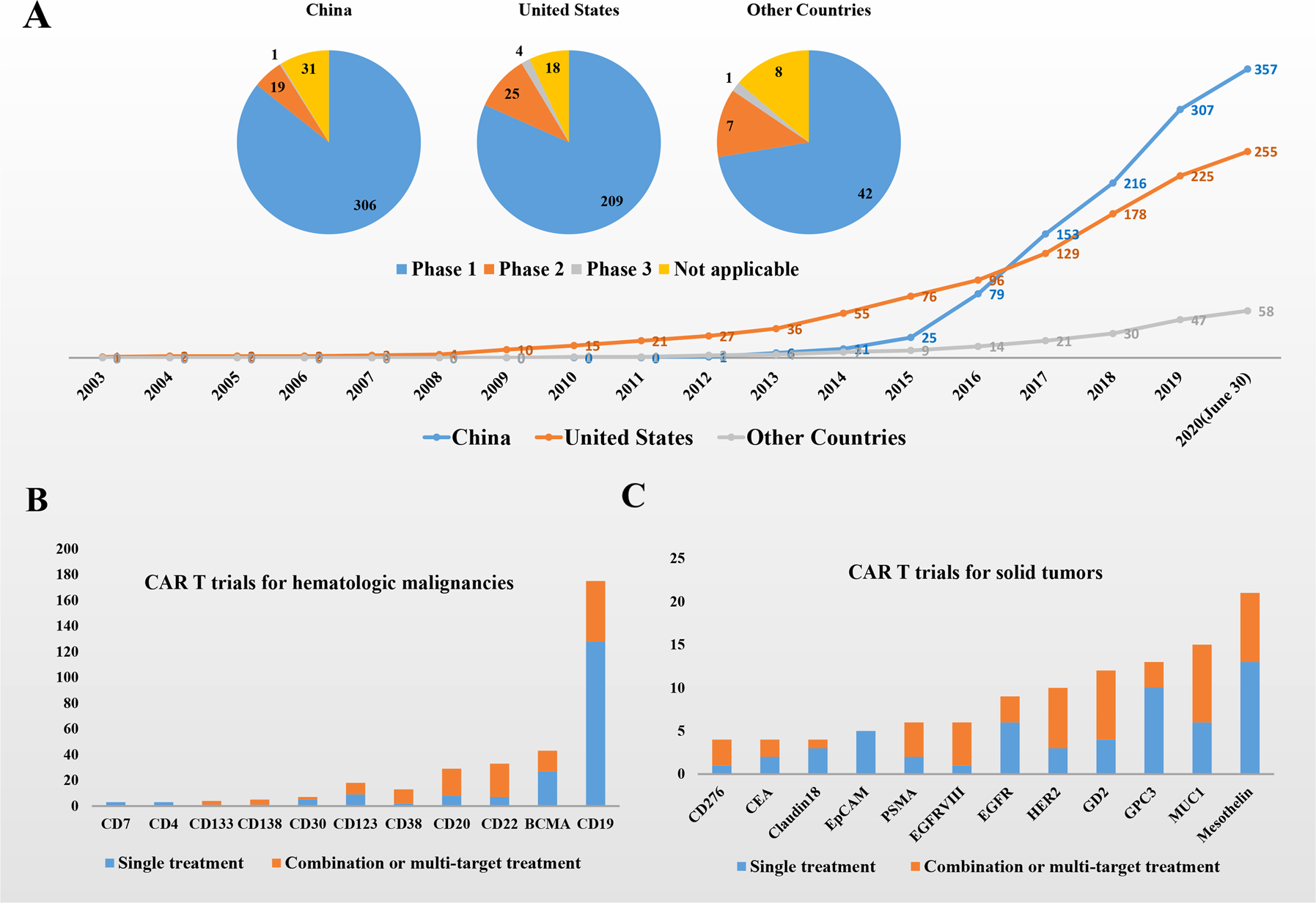
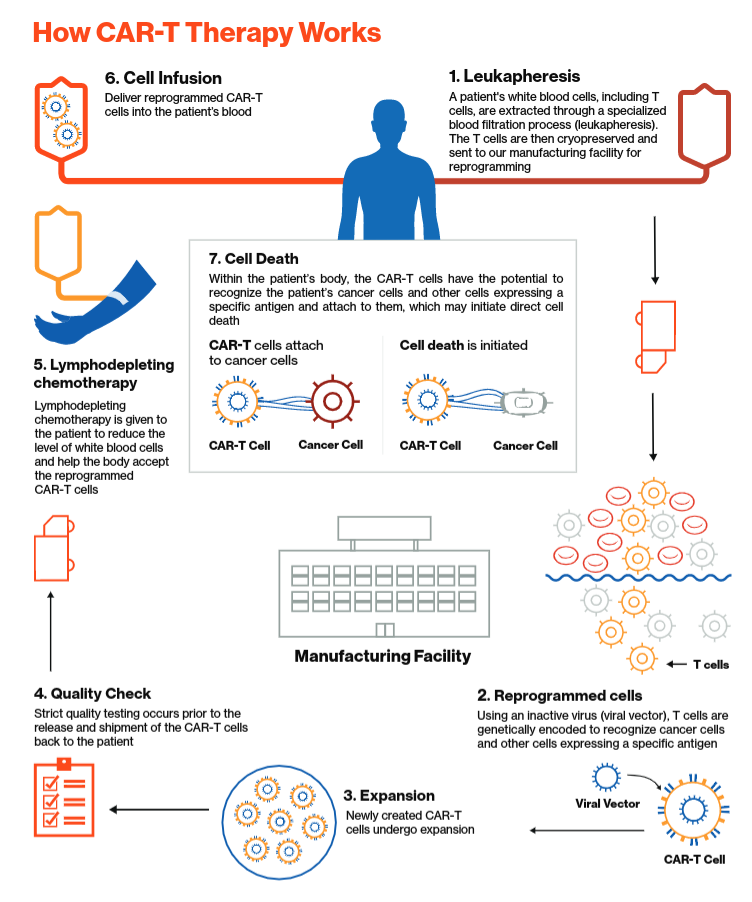
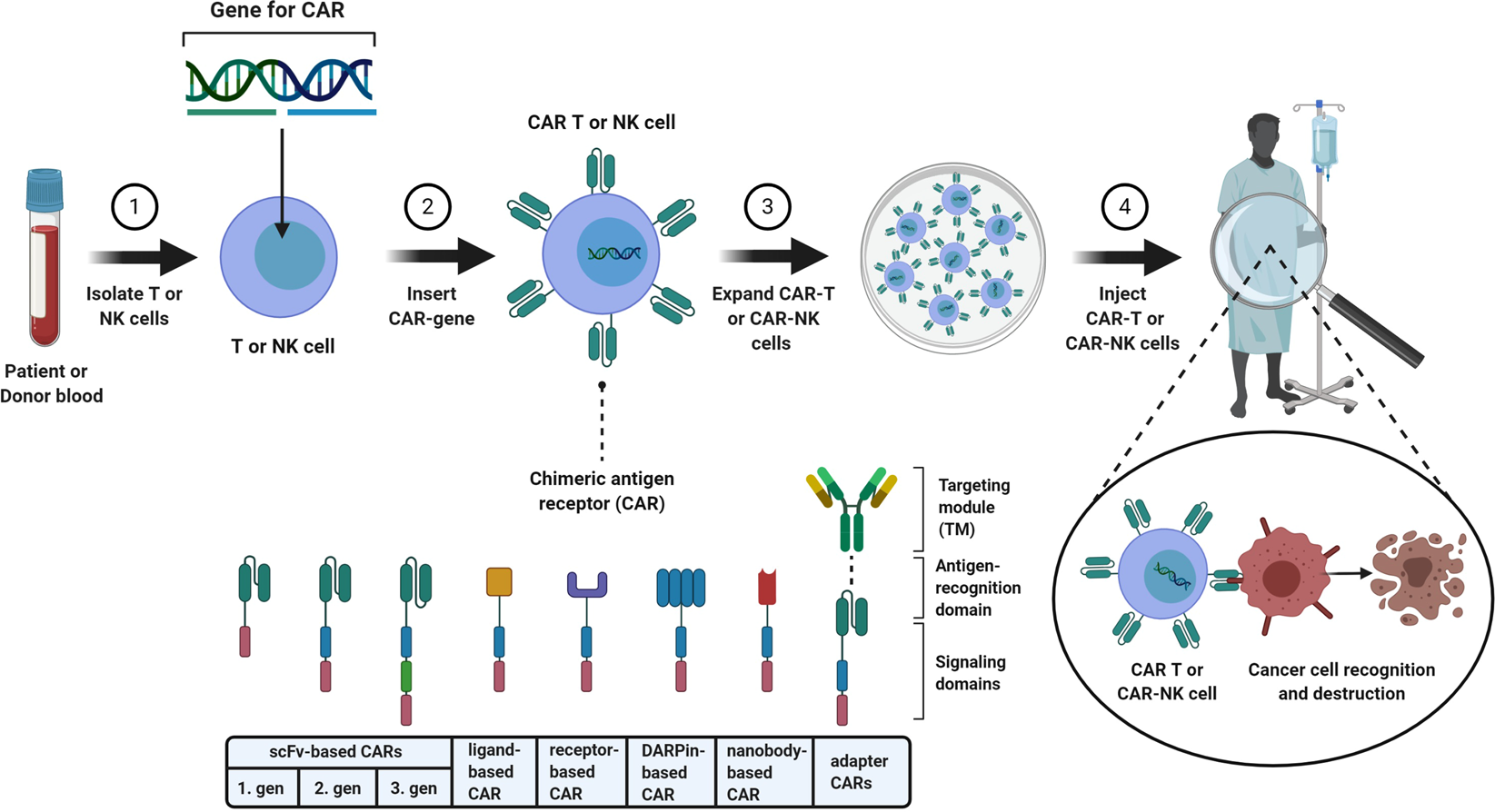
In the News: CTL019 Wins FDA Panel Support, Driving with ADHD, High School Football, New Genetic Syndrome, Teen Bone Growth, Missed Nursing Care

Novartis Announces NEJM Publication Of Updated Analysis From ELIANA Trial Showing Longer-Term Durable Remissions With Kymriah™ In Children, Young Adults With r/r ALL

CD19-targeted chimeric antigen receptor T-cell therapy for CNS relapsed or refractory acute lymphocytic leukaemia: a post-hoc analysis of pooled data from five clinical trials - The Lancet Haematology

Novartis' CTL019 Applied for Clinical Trial in China to Officially Enter the Chinese Market |_Pharmasources.com

Optimizing CAR-T Cell Manufacturing Processes during Pivotal Clinical Trials: Molecular Therapy - Methods & Clinical Development
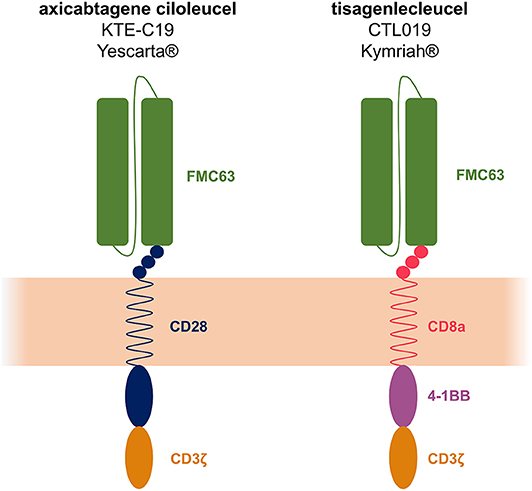
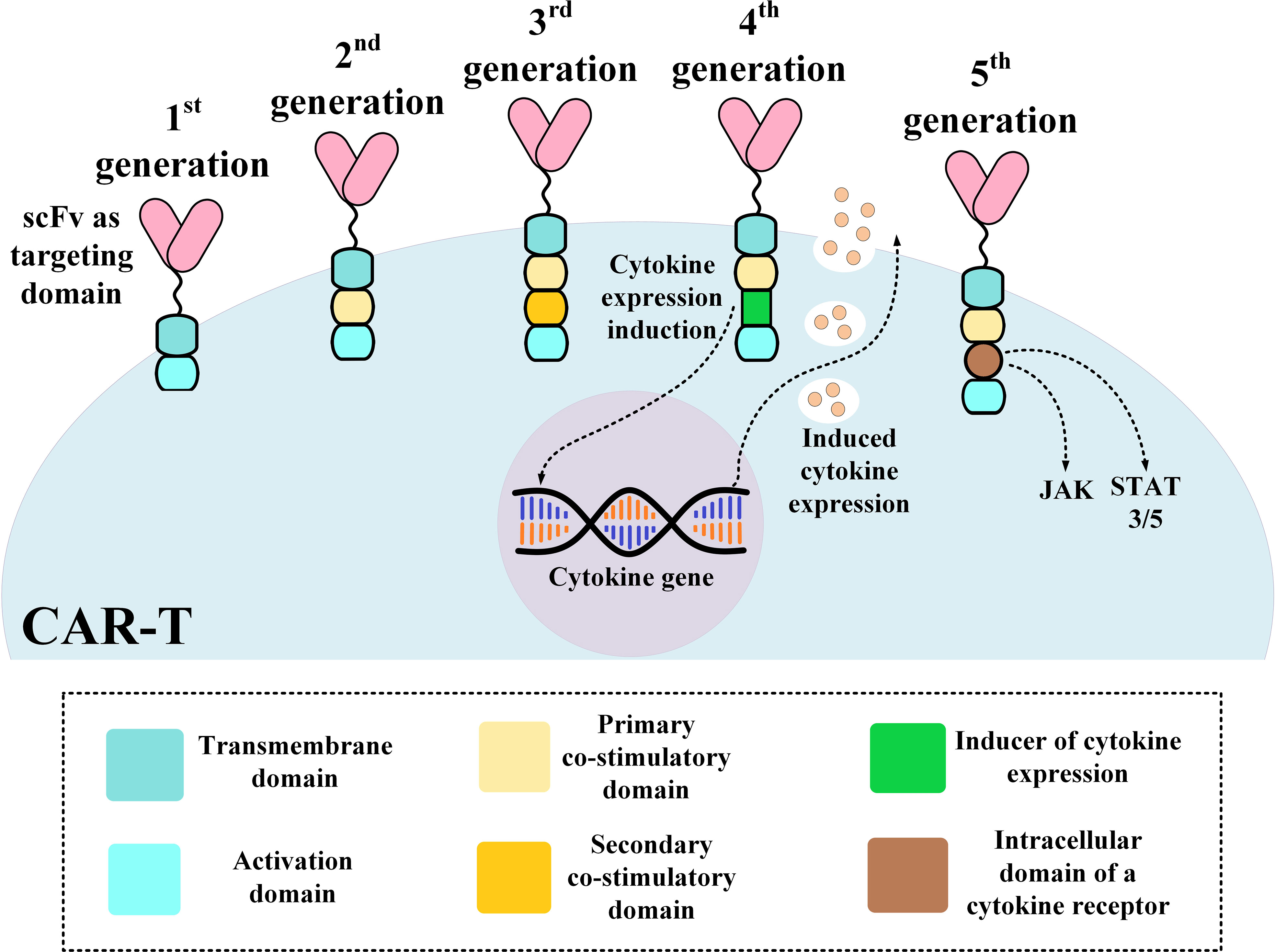
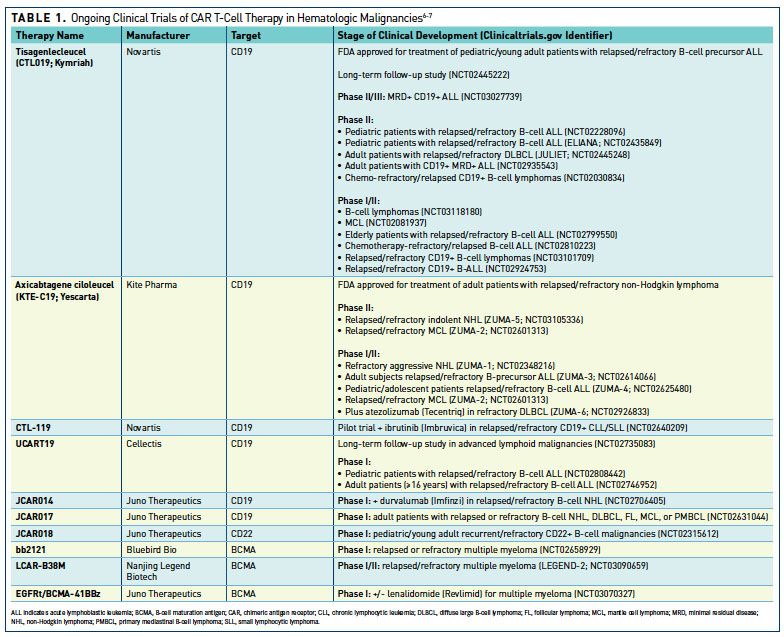
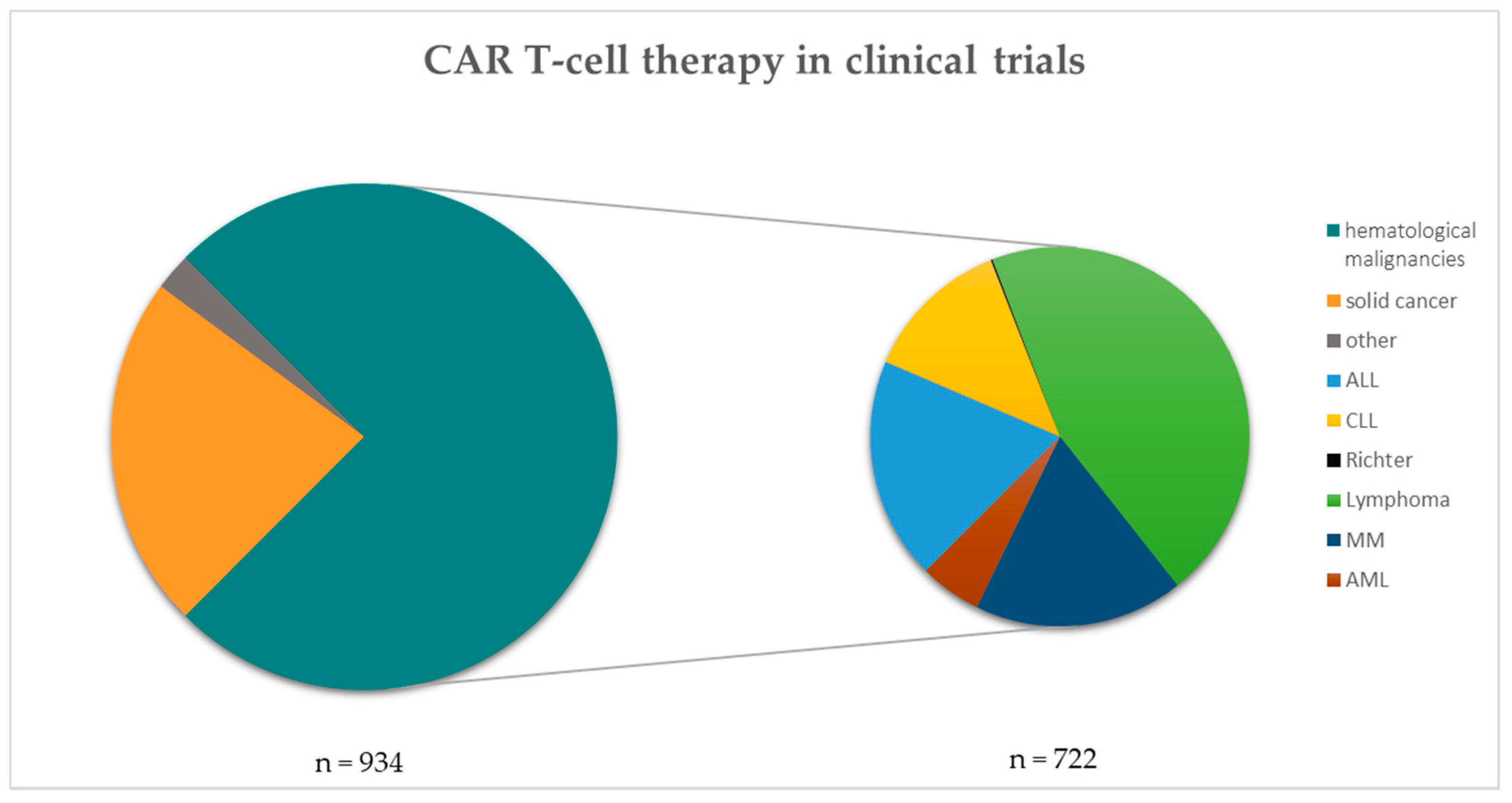
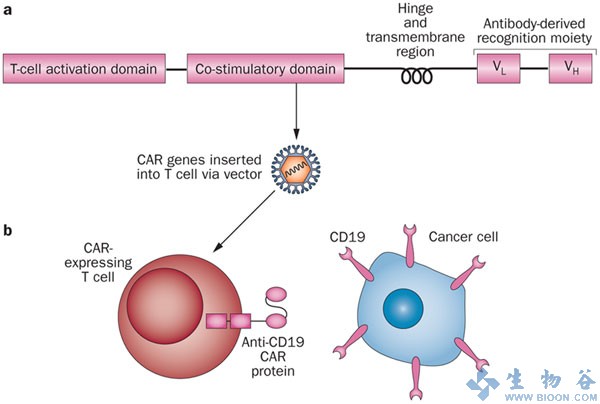
Frontiers | The Advent of CAR T-Cell Therapy for Lymphoproliferative Neoplasms: Integrating Research Into Clinical Practice | Immunology