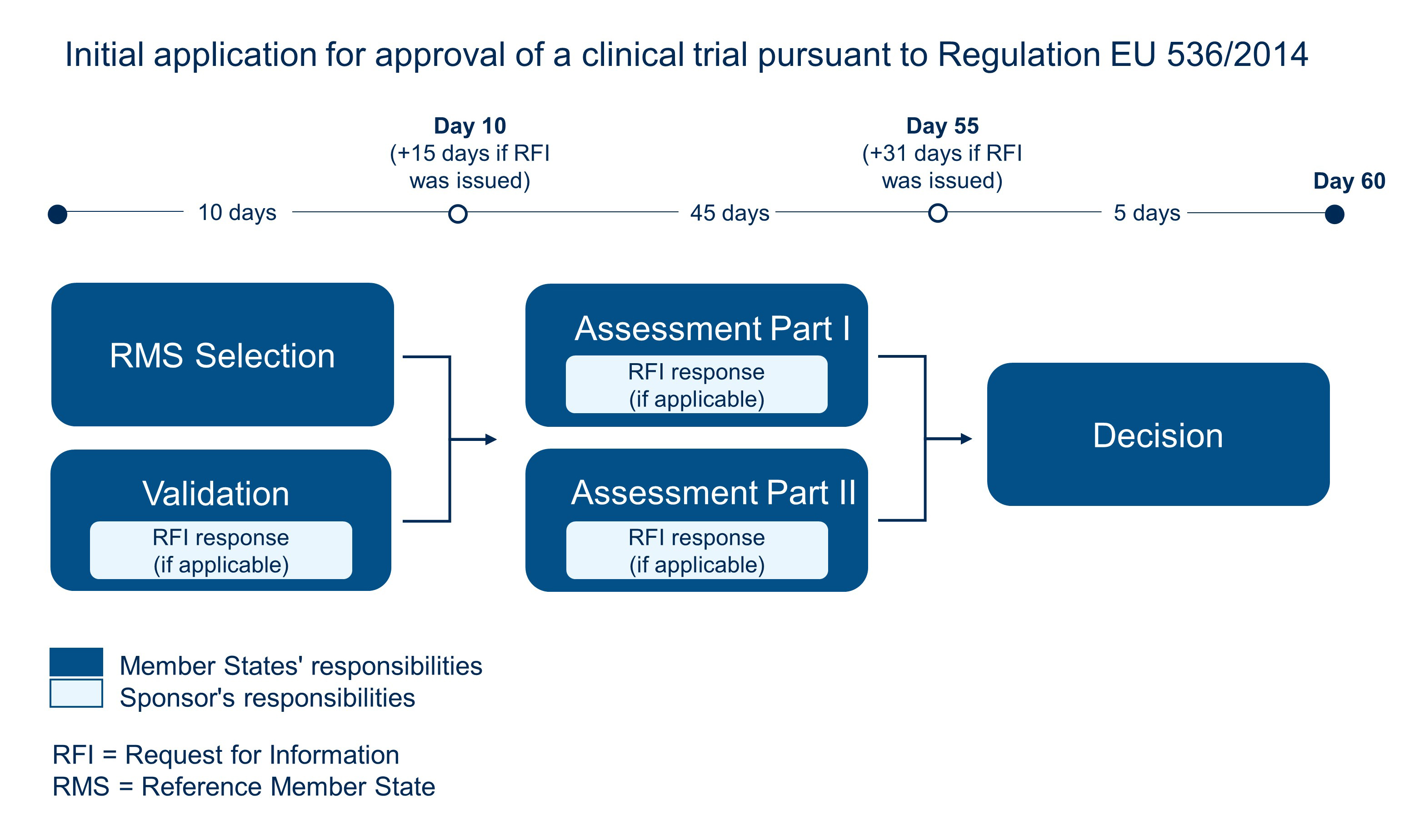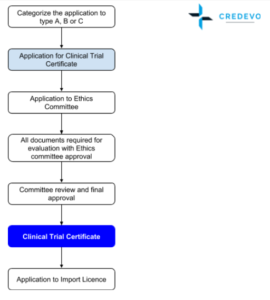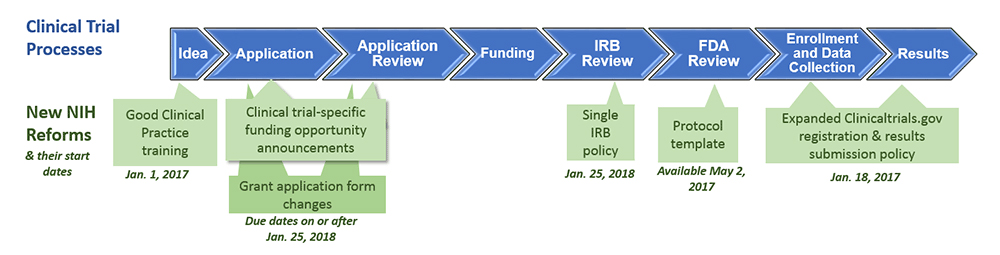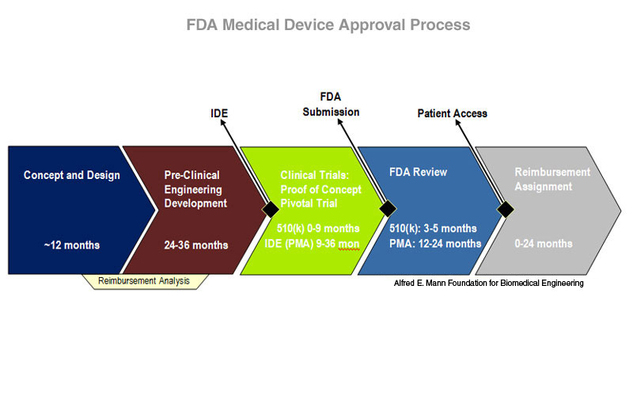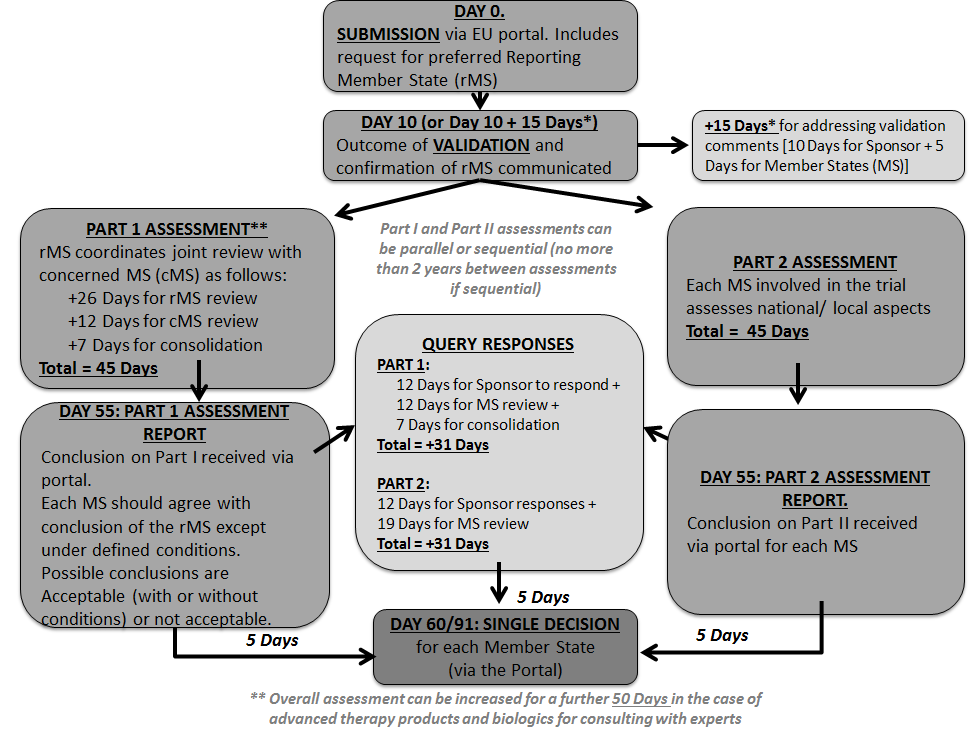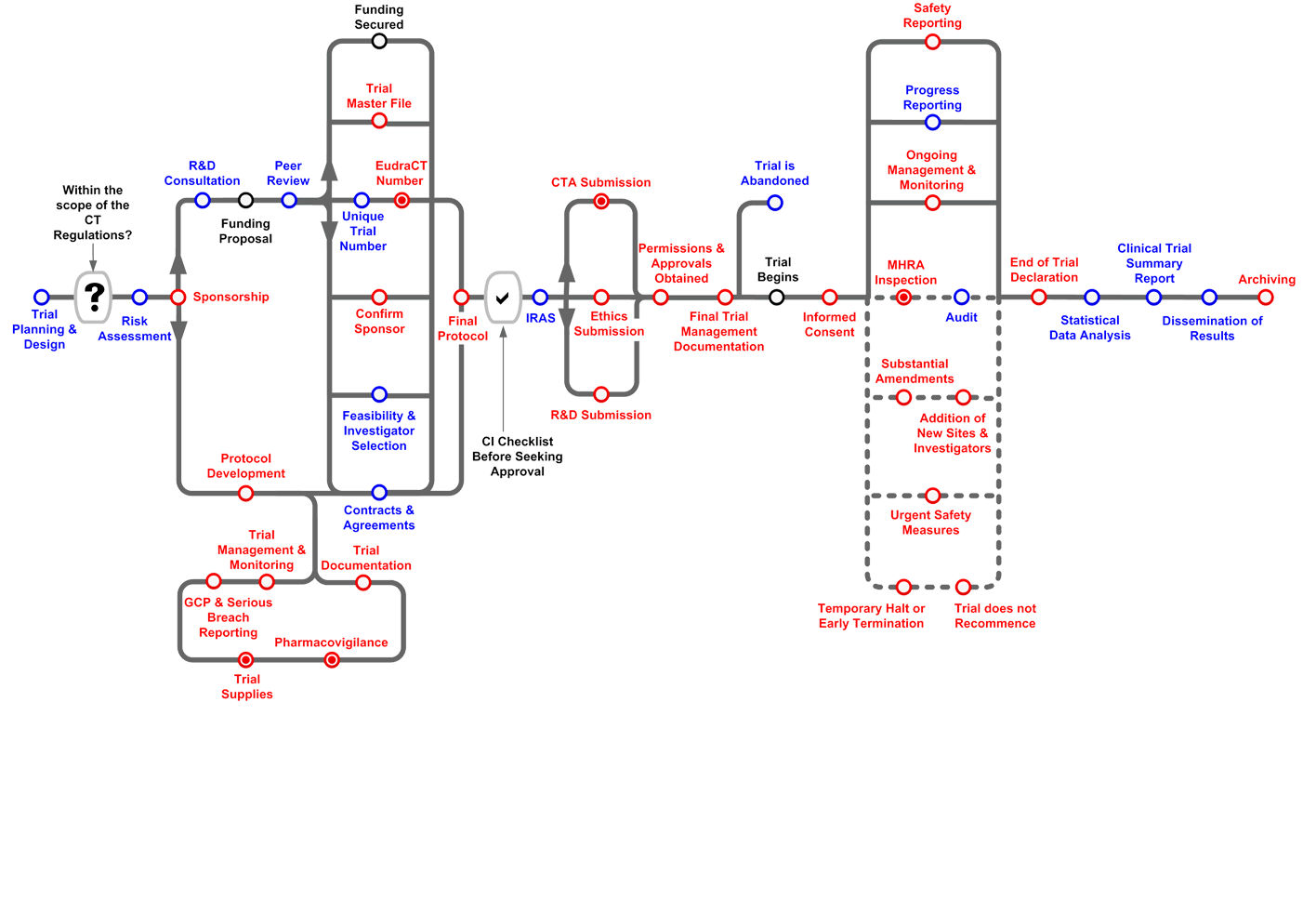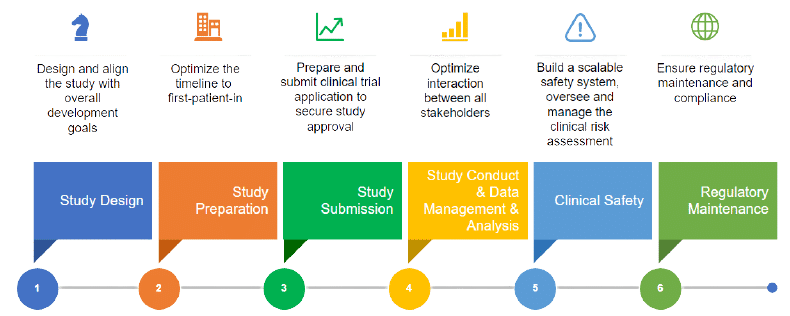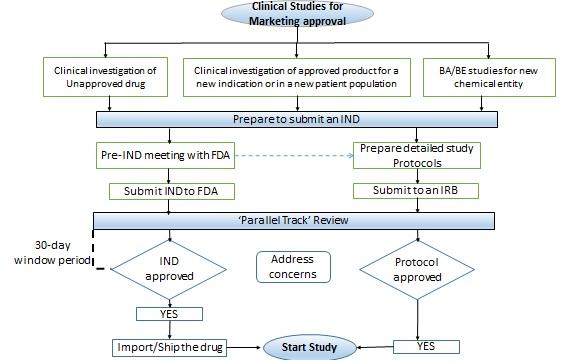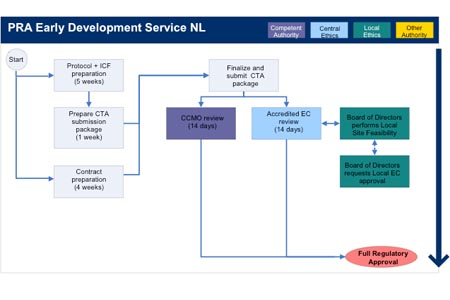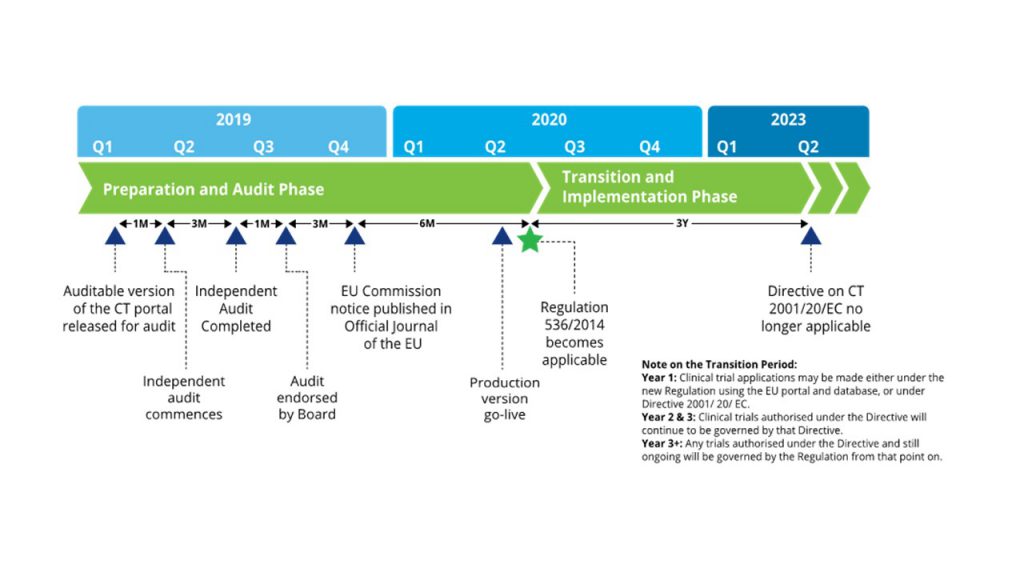
ADVANCED CLINICAL TRIALS THE CLINICAL TRIAL PROCESS: IMPENDING CHANGES IN THE REGULATORY FRAMEWORK - ADVANCED CLINICAL TRIALS

Indicative clinical trial application review process in India, which... | Download Scientific Diagram
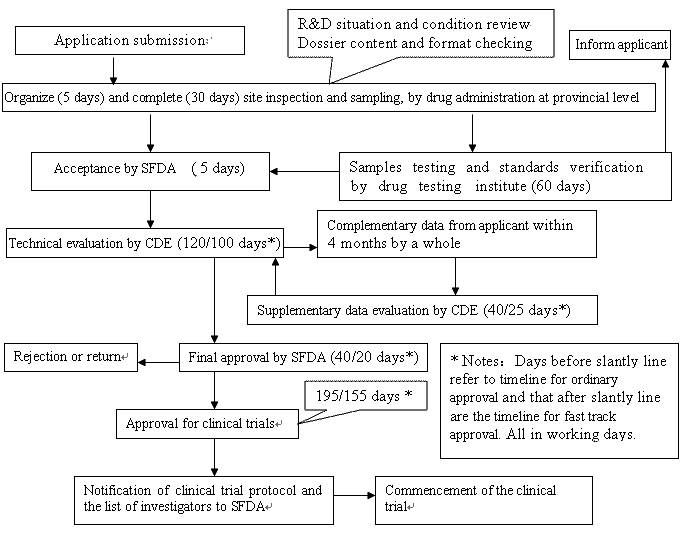
Application and approval procedure for clinical trials-China FDA,SFDA,CFDA,MOH,MOA,AQSIQ,CNCA,CIQ registration approval license for cosmetics,health food supplement,medical device,IVD,drug,infant milk powder,dairy,pet food ,disinfectant etc.

Regulatory Approval – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India
Current clinical trial approval process in India. Abbreviations: BM,... | Download Scientific Diagram

File:Figure 1 Food and Drug Administration's (FDA) Typical Drug Development and Approval Process (35856478702).jpg - Wikimedia Commons

New regulation on clinical trials in Spain - Leon Research | CRO - Clinical Trials Spain, Italy and Portugal

Regulatory Approval – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India