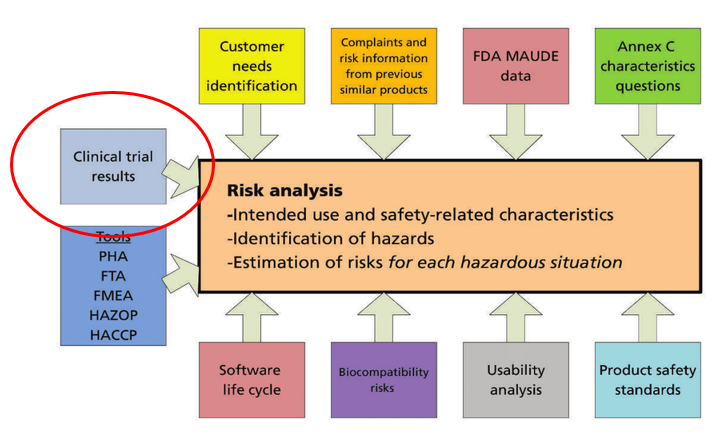
White Paper Clinical Evaluation of Medical Devices: The Increasing Responsibilities of Clinical Evaluators
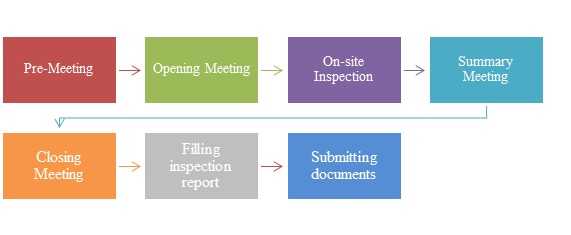
On-site Inspection Procedure of Medical Device Clinical Trials in China - Regulatory News - Medical Devices - CIRS Group

Regulation of Medical Devices and their Clinical Trials Studies in the USA: An Update | Bentham Science

Clinical Evaluation and Investigation of Medical Devices under the new EU-Regulation by Wolfgang Ecker, Gerold Labek, Tarquin Mittermayr - Ebook | Scribd
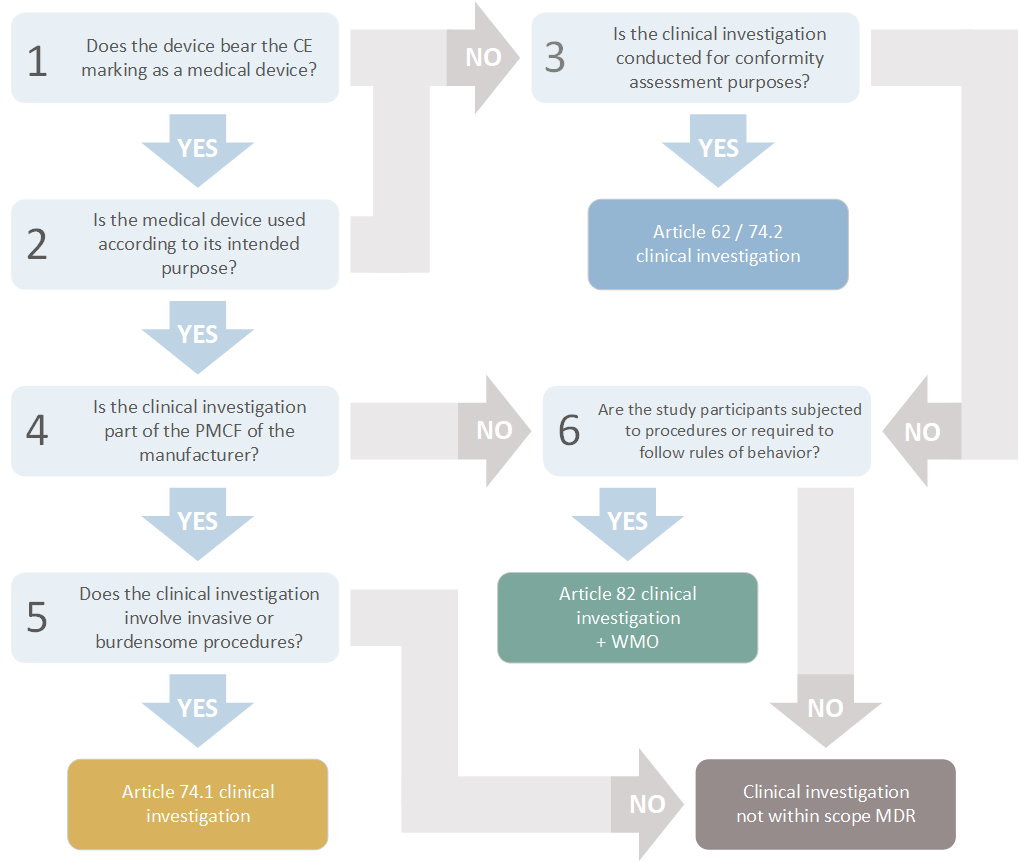
Clinical investigations: definition and framework | Investigators | The Central Committee on Research Involving Human Subjects

MDR - Medical Device Regulation. Regulation of the EU- European Union on the Clinical Investigation and Sale of Medical Devices Stock Vector - Illustration of healthcare, regulate: 212157525

Medical device regulation in Europe – what is changing and how can I become more involved? - EuroIntervention